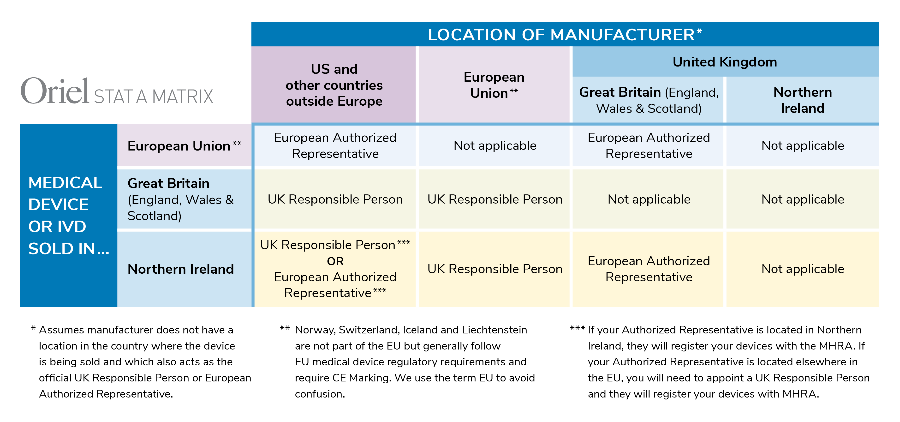
ABPI-BIA submission to MHRA consultation on EU Exit 'no deal' contingency legislation for the regulation

The European Commission's key messages on the EU budget 2020 discharge | CDE Almería - Centro de Documentación Europea - Universidad de Almería

European Regulatory Roundup January 2022: EU's Year Gets Off To Highly Productive Start :: Medtech Insight
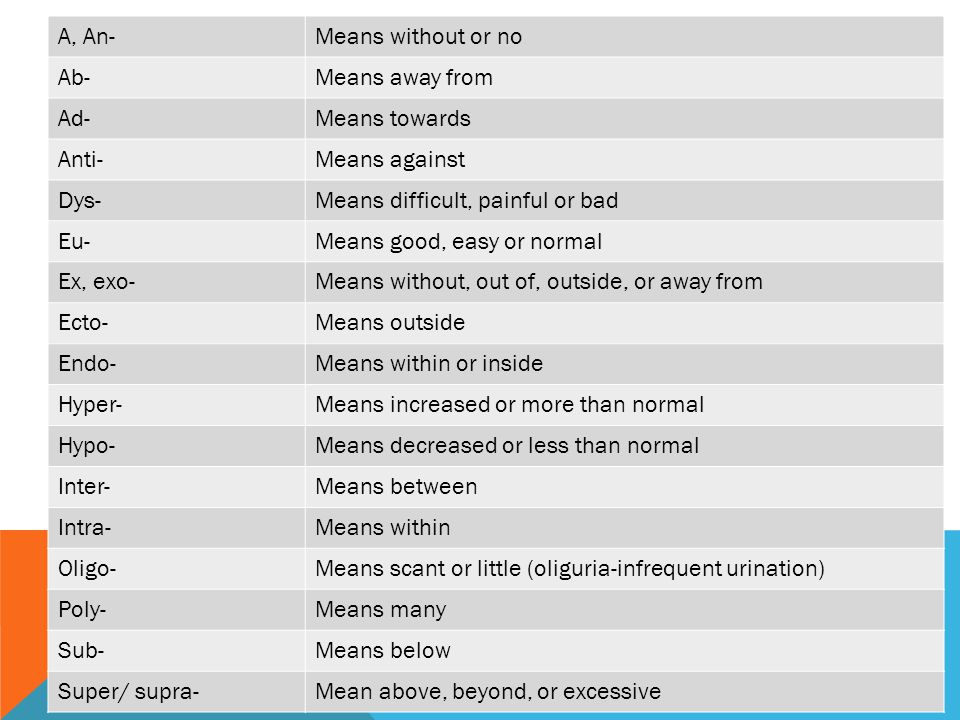






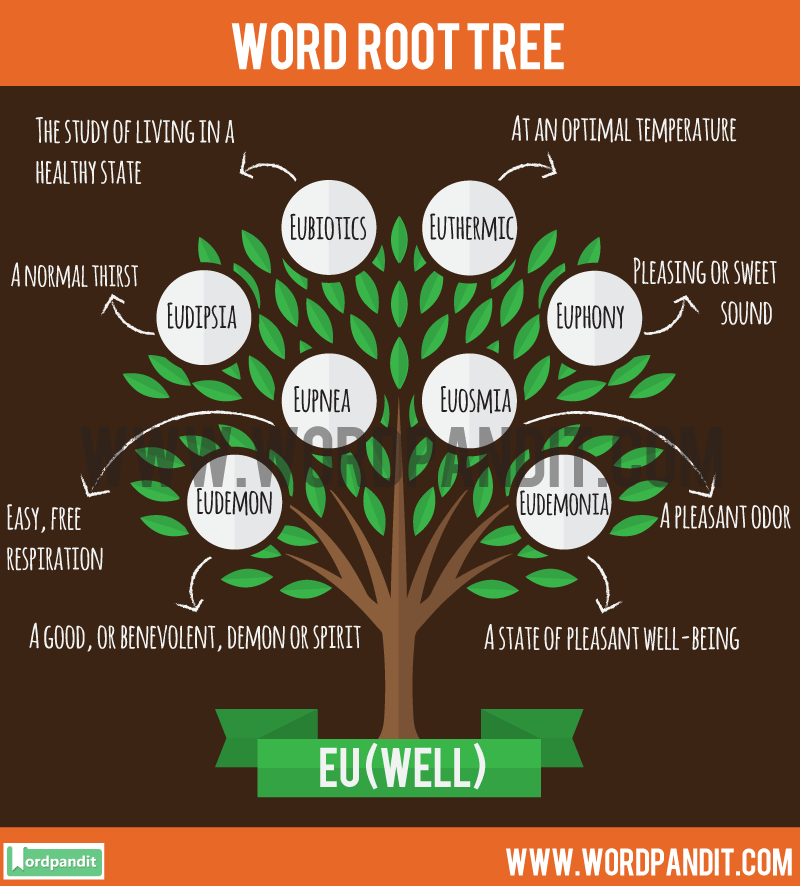
![PDF] A glossary of terms related to complementary and alternative medicine | Semantic Scholar PDF] A glossary of terms related to complementary and alternative medicine | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e58e6233eced2318866be3ff99a4b095a961f7c8/28-Table1-1.png)