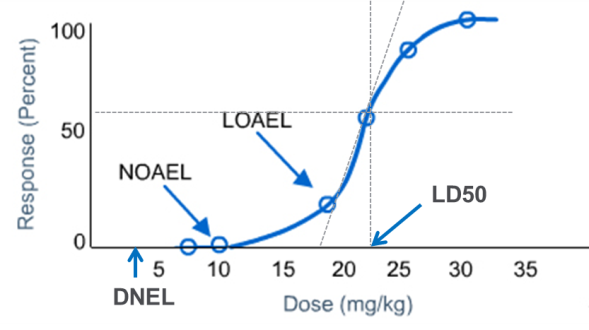
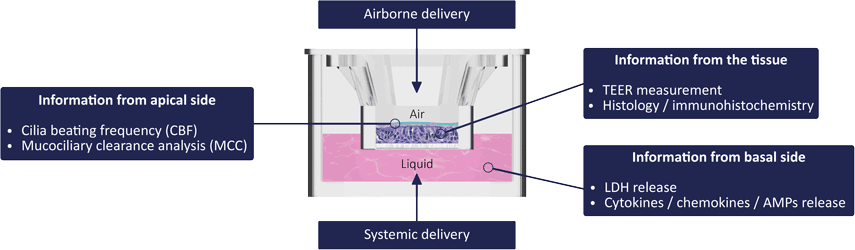
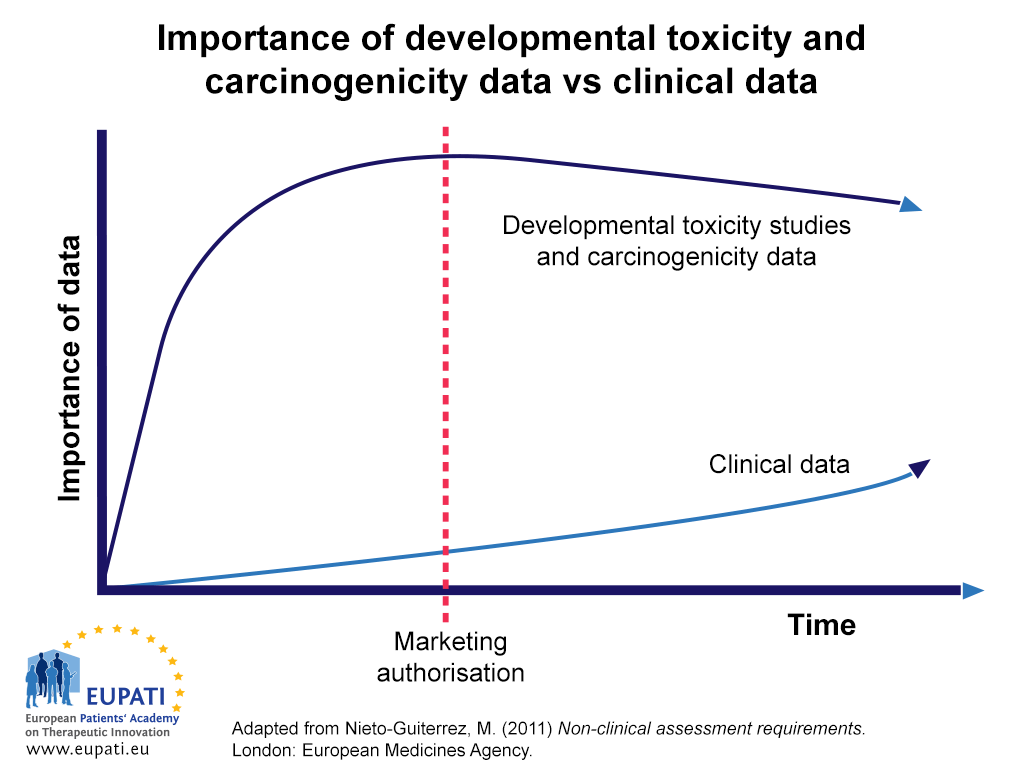
An Integrated Decision-tree Testing Strategy for Repeat Dose Toxicity with Respect to the Requirements of the EU REACH Legislati

Scheme of repeated dose toxicity study for VGX-6150. C57BL/6 mice were... | Download Scientific Diagram

Environmental Health II. Toxicology Shu-Chi Chang, Ph.D., P.E., P.A. Assistant Professor 1 and Division Chief 2 1 Department of Environmental Engineering. - ppt download

Acute vs. Chronic Toxicity: Differences & Overview | What is Toxicity? - Video & Lesson Transcript | Study.com

Molecules | Free Full-Text | Single, 14-Day, and 13-Week Repeated Dose Toxicity Studies of Daily Oral Gelidium elegans Extract Administration to Rats